|
|
Anesthesia Pharmacology Chapter 33: Overview of Chronic Obstructive Pulmonary Disease (COPD)
COPD is a major category of OVD and within the COPD category is found the following diseases: chronic asthma, chronic bronchitis, and emphysema.
Asthma is considered a chronic disorder even though during remission, patients may appear completely normal with no symptoms. However, because asthma has a chronic, inflammatory components transition from the relatively increased bronchomotor tone to a significantly increased bronchomotor tone that can abruptly diminished pulmonary function, asthma is considered a COPD.
In the general case, COPD is characterized by airflow obstruction which may be intralumenal or extralumineal, but in either case air trapping occurs.
The intralumenal small airways may also be changed by abnormal ciliary function, ineffective secretion clearanceas well as increased mucus production.
The process of aspiration from the pharynx or stomach as well as the underlying, in some cases, chronic inflammatory process contribute to increased airway secretion.
Airway wall changes, particularly in asthma and bronchitis include:
bronchiolar smooth muscle hyperreactivity and proliferation (hypertrophy). Decreases in luminal diameter manifest as exhalation airflow obstruction accompanies airway inflammation and edema.
Extralumineal airway changes manifesting as airway obstruction can be due to mass effects which directly compress the airway and include effects of tumors or lymph nodes. Other factors include lung parenchyma loss with associated structural support loss as well as significant edema. All these effects may occur to a greater or lesser extent in the COPD patient.
Emphysema has been categorized as type A COPD, presenting in "pure" form, but sometimes presenting a more mixed or ambiguous clinical picture in which elements of chronic bronchitis may also be present.
The pathophysiological characteristic of emphysema follows from lung parenchymal loss as well as reduced effect of surface area for gas exchange. The latter aspect is due to loss of "radial traction" within the alveoli.
Patients with emphysema are typically acyanotic and exhale through pursed lips which has the effect of providing end-expiratory pressure.
Arterial oxygen tension may be maintained despite relatively advanced emphysema. Only rather late in the disease trajectory, will changes (increases) in alveolar and arterial CO2 tension the observable since typically patients will not retain CO2.
|
|
|
|
|
|
|
|
|
19Normal lung chest X-ray (left) compared to chest X-ray from a patient with emphysema:
"The findings of hyperinflated lungs, flattened diaphragms, diminshed vascular markings suggest emphysema.
Emphysema destroys alveoli and results in obstruction of small airways leading to air trapping - increased lung volumes.
On lateral film there may be increased retrosternal space.
However, thin individuals with good inspiratory effort may have flattened diaphragms and hyperinflated lungs that could mimic emphysema on cxr."
|
|
20"PA (left) and lateral (right) radiographs of a young woman with alpha-1-antitrypsin deficiency and typical changes of severe obstructive lung disease. There is diminished vascularity (arterial deficiency pattern) along with flattened diaphragms and an increase in the AP dimension of the chest."
1In emphysema, spirometry reveals a characteristic signature, primarily a decrease in FEV1/FVC ratios. This ratio reduction is a major feature of chronic airflow obstruction.
Furthermore in emphysema there will be reduced airflow at all lung volumes.
Lung hyperinflation with air trapping will be reflected in an increase in the ratio of RV (residual volume)/TLC (total lung capacity).
Other characteristics include low, flat diaphragms with hyperlucent lung fields (see radiograph above) due to air trapping as well as loss of lung tissue.
Emphysema patients usually do not present with right heart failure or cor pulmonale and tend not to have repeated pulmonary respiratory failure events.
1Chronic bronchitis is categorized as type B COPD and pathologically reflects changes consistent with hypertrophy and bronchial goblet cell proliferation.
Furthermore hyperreactivity is associated with the bronchial smooth muscle.
Changes in goblet cell physiology result in increased mucus secretion. This significant secretion may occlude airways.
Cough is typically present and the cough is productive and chronic; furthermore, secretions promote infection secondary to gram-positive and gram-negative bacterial growth.
|
1Chronic bronchitis patients exhibit typically reduction in arterial oxygen tension with desaturation occurring early in the disease trajectory. These effects are in distinction to those observed in emphysema patients. The alveolar and arterial CO2 can increase in bronchitis resulting in hypercapnia and hypoxia. Arterial oxygen the saturation with attenuated cardiopulmonary function result in patients appearing cyanotic, bloated, and plethoric ("a bodily condition characterized by an excess of blood and marked by turgescence and a florid complexion" ).
By contrast emphysema patients who might be called "pink puffers" [because they tend to puff, through pursed lips and they tend to be well oxygenated, hence pink] bronchitis patients might be characterized as "blue bloaters" [because they exhibited relatively poor oxygenation, hence blue and are bloated in appearance; this physical appearance characterization occurs in part because chronic bronchitis patients may have over the years withdrawn from physical activity and may be overweight with poor muscle tone.].
1Resting pulmonary function exhibits a reduced FEV 1/FVC ratio (similar to emphysema).
However lung volumes remain typically near-normal except during a worsening period induced by infection or bronchospasm.
Relative to baseline lung volumes, reduced volumes can be documented radiographically.
Respiratory compromise (and sometimes failure) may be associated with chronic bronchitis disease evolution. A worsening of the disease state would be associated with increased frequency of bronchospasm with pulmonary infection as well as increased mucus secretion.
During a disease exacerbation episode, the hypoxic and hypercapnic state will be more pronounced. Despite this condition, very ill patients exhibiting hypoxia and hypercapnia probably should not be intubated since tracheal intubation with mechanical ventilation appear poorly tolerated. Alternatively, low-flow oxygen administration with pharmacological intervention is probably more appropriate for management of the exacerbated state.
1Chronic bronchitis patients will also exhibit, secondary probably to reduced arterial oxygen saturation, increased pulmonary vascular resistance. With disease progression, this increased pulmonary resistance promotes right ventricular hypertrophy and ultimately may lead to right-side heart failure and cor pulmonale. Systemic manifestations include peripheral edema and hepatojugular reflux.
The underlying pathology in asthma is chronic inflammation.
Associated with the state is bronchial hyperreactivity and asthma itself is characterized by relatively long apparently asymptomatic periods with occasional acute exacerbations.
Even during the asymptomatic period, pulmonary function tests may be abnormal, although not necessarily. However, during the exacerbation, with increased bronchiolar smooth muscle tone, the airway becomes constricted and airflow obstruction evident.
In this case, changes in FEV 1/FVC ratio are apparent. The thickness of secretions during an acute exacerbation further impair exhalation airflow.
Changes in bronchiolar smooth muscle tone may involve alterations in a balance between factors that modulates smooth muscle generally; for example, cAMP and cGMP.
Other modulating factors include immunoglobulin E (IgE). The details in terms of the sequence of events associated with asthma are described in a separate section.
Bronchiolar smooth also tone is also influenced by
the activity of the autonomic nervous system, given that sympathetic
and parasympathetic input results in differing effects. Parasympathetic
system activation tends to promote smooth muscle contraction (bronchoconstriction);
by contrast, sympathetic activation promote smooth muscle relaxation.
This principle explains of course why
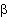 2
adrenergic receptor agonists on one hand an an antimuscarinic drug
ipratropium on the other are effective drugs in asthma
management.
2
adrenergic receptor agonists on one hand an an antimuscarinic drug
ipratropium on the other are effective drugs in asthma
management.
 In anesthesia,
manipulation and intubation of the upper airway may provoke
bronchospasm. In accord with the preceding analysis, antimuscarinic
agent administration would be reasonable in such a patient.
In anesthesia,
manipulation and intubation of the upper airway may provoke
bronchospasm. In accord with the preceding analysis, antimuscarinic
agent administration would be reasonable in such a patient.
1Pulmonary Obstruction and COPD
As noted above, initially for emphysema and chronic bronchitis disease processes, FVC is normal (initially). Airflow obstruction in both cases is associated with a reduced maximal voluntary ventilation value (MVV). Diagnosis of these conditions would depend on the history, physical examination as well as demonstration by pulmonary function testing (educed FEV1/FCC ratio).
Lung volume measurements in patients with chronic bronchitis will typically be normal except during an acute disease exacerbation. On the other hand, in patients with emphysema will exhibit an increase in TLC and a more substantial increase in functional residual capacity (FRC) and residual volume (RV). The RV/TLC ratio or the FRC/TLC ratio effectively identify emphysema-related hyperinflation with gas trapping.
In chronic bronchitis, carbon monoxide diffusion capacity is approximately normal since the disease itself is centered and bronchial smooth muscle rather than lung parenchyma. By contrast, in emphysema, loss of parenchymal surface area supporting gas exchange manifests as a reduced carbon monoxide diffusion capacity.
As either disease progresses, each may manifests characteristics of the other.
One example is that in a patient who has chronic bronchitis, altered lung volume may be noted and diffusion capacity could appear similar to a patient with emphysema. Severe emphysema patients (significant ventilatory compromise with hyperinflation), moreover, may exhibit reduced vital capacity because of lung volume changes and when this happens, a prolongation in FEV1 with a decrease in FVC would appear consistent with hyperinflation.
The asthmatic patient will tend to exhibit specific pulmonary function anomalies particularly during periods of disease worsening (exacerbations).
A reduced FEV1/FCC ratio as well as airflow reduction throughout the range of vital capacities are noted. Specific changes are described in terms of maximal midexpiratory flowrate (MMEFR), maximal midflow rate (MMFR) and FEF 50.
During bronchospasm, the RV/TLC and the FRC/TLC ratios can increase.
Asthmatic patients often respond to bronchodilator
drugs which will alter spirometric data. Airflow improvement and
even increased vital capacity may be observed following
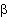 2
agonist administration.
2
agonist administration.
The extent of airflow improvement following the administration of such bronchodilators may vary, but a clinically significant response corresponds to about a 20%-25% FEV1 or FVC increase.
Gas exchange abnormalities are predicted as pulmonary mechanics become abnormal.
Bronchospastic disease changes gas flow and blood distribution possibly causing, in some cases, ventilation-perfusion mismatching. With bronchospasm, autoregulation mechanisms change blood flow patterns in an attempt to maintain a match between ventilated regions and perfused regions. Nevertheless, chronic bronchitis and acute asthma often result in a low ventilation/perfusion condition (V/Q) with associated oxygen desaturation and hypoxemia. Use of bronchodilator intervention improves ventilation/perfusion relationships by reducing airflow obstruction, promoting gas exchange, and reducing perfusion anomalies.
An initial reduction in oxygen saturation may occur following bronchodilator treatment because of initial effects on vascular smooth muscle (vasodilation) prior to the effect on bronchiolar smooth muscle (bronchodilation). The initial vasodilation increases blood flow through poorly oxygenated lung parenchyma, thus initially resulting in reduced oxygen saturation. Eventually, the net effect is improvement in oxygen saturation which occurs very shortly after the initial transient.
Acute bronchospasm is associated with potentially severe hypoxemia, although CO2 elimination is normal. This results is consistent with linearity exhibited by the CO2 dissociation curve, by contrast to the sigmoidal oxyhemoglobin dissociation curve.
|
|
Note the relative linearity of the CO2 saturation curve compared to the O2 dissociation curve. Hyperventilation can thus lower CO2 tension and content in a nearly linear manner; however, increasing O2 tension does not have much effect on O2 content in the normal (middle) range because of the sigmoidal shape. Sigmoidicity reference the positive cooperatively exhibited by hemoglobin-O2 binding isotherms.
Accordingly, CO2 elimination maintains near normal until V/Q mismatching and bronchospasm proceed to an extremely severe extent. Therefore, if arterial CO2 tension begins to rise in this patient category (asthmatic), overt respiratory failure may be imminent.
In patients with chronic bronchitis, chronic CO2 retention develops very slowly and accounts for the compensating respiratory acidosis characteristic of arterial blood gas data in this patient group. The increase in carbon dioxide tension may be due to increased breathing work which presents late in the disease course. Eventually, compensatory mechanisms may not be sufficient to maintain a normal CO2 tension.
Patient with chronic bronchitis will also respond to bronchodilator administration due to the presence of a bronchospastic component. The magnitude of the improvement is typically substantially less than that which would be noted in the asthmatic patient (about 5%-10% versus 25% improvement (asthma; spirometric data).
 The value of
spirometric testing with respect to bronchodilator responsiveness is
clear in identifying those preoperative patients who may benefit by
preoperative bronchopulmonary dilator drugs. Pharmacologically
induced and sustained airflow increases probably improve both
intraoperative and postoperative courses. This approach probably
decreases morbidity and mortality especially for patients undergoing
upper abdominal operations.
The value of
spirometric testing with respect to bronchodilator responsiveness is
clear in identifying those preoperative patients who may benefit by
preoperative bronchopulmonary dilator drugs. Pharmacologically
induced and sustained airflow increases probably improve both
intraoperative and postoperative courses. This approach probably
decreases morbidity and mortality especially for patients undergoing
upper abdominal operations.
1Bronchodilator Pharmacological Treatment
1, 23
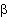 2
agonist treatment is a mainstay for management of bronchospasm.
2
agonist treatment is a mainstay for management of bronchospasm.
The underlying molecular effect may be to increase levels c AMP which ultimately affects the phosphorylation state of myosin light chains.
Examples of drugs that are commonly use sympathomimetic agents to accomplish this goal include metaproterenol and albuterol.
Another drug group useful in promoting bronchodilation is the methylxanthines including aminophylline (85% theophylline).
The basic mechanism of action of aminophylline and other methylxanthines remains to be determined.
One possibility is that these drugs inhibit phosphodiesterase, an enzyme that degrades cyclic nucleotides such as cAMP; however, although such action would explain cardiac stimulatory effects, it is unclear that sufficiently high concentrations of drug required to inhibit phosphodiesterase can in fact be reached in vivo.
Another possibility, with respect to bronchodilation, is that methylxanthines, inhibit access of adenosine to its cell surface receptor. Adenosine itself promotes bronchoconstriction.
However, some methylxanthines which do not inhibit adenosine from binding to its receptors turn out to be more effective in inhibition all bronchoconstriction.
In terms of skeletal muscle mechanics, methylxanthines appeared to strengthen contraction of skeletal muscle which may reverse diaphragmatic fatigue in patients with lung disease.
|
|
Other drugs outside of the sympathomimetic class include parasympatholytics agents, most prominently, ipratropium. Antimuscarinic drugs blocking the effect of acetylcholine at muscarinic receptors reduce bronchiolar smooth muscle tone, normally enhanced by tonic acetylcholine release.
Meter dose inhalers are used for
administration of both
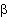 2
agonist drugs and antimuscarinic agents in this application.
Some drugs used for prophylaxis include the corticosteroids and
cromolyn sodium.
2
agonist drugs and antimuscarinic agents in this application.
Some drugs used for prophylaxis include the corticosteroids and
cromolyn sodium.
Drugs useful in management of bronchospasm are discussed in the section on asthma.
1Preoperative Evaluation Overview:
Preoperative evaluation in patients with possibly compromised pulmonary function typically should include baseline spirometry administered in a way to assess pre-and post-bronchodilator effect. Additionally, the maximum volume ventilation or MVV should be determined.
The chronically hypoxic or hypercapnic patient can be identified and characterized by arterial blood gas data.
Particularly helpful for patients about to undergo thoracotomy are tests which measure lung volumes and diffusing capacity. This type of additional testing is unlikely to be valuable for those undergoing abdominal operations and it is unclear whether extended testing is helpful for patients who are elderly, obese or who smoke.
Categories of patients who probably should undergo preoperative evaluation of pulmonary function include:
thoracic surgery patients
upper abdominal surgery patients
heavy smokers with cough
elderly patients (70 years of age)
patients with a documented pulmonary disease history.
1Reduced lung volume (FRC and TLC), increased respiratory rate, reduced tidal volume, an increase in the alveolar-to-arterial oxygen tension gradient are observed in the postsurgical patients often secondary to pain-related splinting and non-pain related abnormal diaphragmatic function [splinting definition: "to protect against pain by reducing the motion of " as in "the patient splinted his chest by a fixed position and shallow breathing "].
The increase alveolar-to-arterial oxygen tension effect noted in the postsurgical patients is probably due to a reduced lung volume (microatelectasis) causing a right-to-left shunting in the lung. These consequences of surgery for can be worsened in the patient in which baseline pulmonary function abnormalities exist.
The net effect of these pulmonary function problems of our noted in the transient restrictive physiological state which exists mainly during the first postoperative day. About 80% of preoperative pulmonary function of these usually observed by the third postsurgical day.
|
|
|
1,25Preoperative Risk Assessment (Pulmonary)
FVC < 50% predicted
FEV1 < 50% predicted of < 2.0 L.
MVV < 50 predicted or < 50L/min
DCO < 50% predicted
RV/TLC > 50%
![]()
1Kopp, VJ and Boysen, PG "Evaluation of the Patient with Pulmonary Disease" Chapter 13, in Principles and Practice of Anesthesiology, 2nd Edition, Longnecker, D.E., Tinker, J.H, and Morgan Jr, G.E., eds), pp 232- 242, Mosby, St. Louis,1998.
Key References Cited by Kopp and Bosen (1):
Celli. B: What is the value of preoperative pulmonary function testing? Med Clin North Am, 7:309, 1993
Crapo, RO: Pulmonary function testing, N Eng J Med 331: 25, 1994
Gracey, DR, Divertie, MB, Didier, EP: Preoperative pulmonary preparation patients with chronic obstructive pulmonary disease, Chest 76: 123, 1979.
Tisi, GN: Pulmonary physiology in clinical medicine, Baltimore, 1980, Williams and Wilkins.
3University of Western Ontario, Department of Pathology; maintained by C.J. Gibson
4Johns Hopkins Interactive Restrictive Physiology, "Restrictive Ventilatory Defect" http://oac.med.jhmi.edu/res_phys/Encyclopedia/RestrictVentDefect/RestrictVentDefect.HTML , Copyright © 1995 Johns Hopkins University
5Pulmonary Alveolar Proteinosis (Ch. 79); Pulmonary Disorders (Section 6); The Merck Manual of Diagnosis and Therapy,
6Michael P. D'Alessandro, M.P and Yasayuki Kurihara,Y.,n Chest X-Ray: Pulmonary Alveolar Proteinosis, University of Iowa
7Department of Pathology, UCSF,
8Johns Hopkins Interactive Restrictive Physiology, "Emphysema" http://oac.med.jhmi.edu/res_phys/Encyclopedia/Emphysema/Emphysema.HTML, Copyright © 1995 Johns Hopkins University
9Johns Hopkins Interactive Restrictive Physiology, "Bronchitis" http://oac.med.jhmi.edu/res_phys/Encyclopedia/Bronchitis/Bronchitis.html, Copyright © 1995 Johns Hopkins University
10Johns Hopkins Interactive Restrictive Physiology, "Asthma" http://oac.med.jhmi.edu/res_phys/Encyclopedia/Asthma/Asthma.html, Copyright © 1995 Johns Hopkins University
11Johns Hopkins Interactive Restrictive Physiology, "Obstructive Ventilatory Defects" http://oac.med.jhmi.edu/res_phys/Encyclopedia/ObsVentDefect/ObsVentDefect.HTML, Copyright © 1995 Johns Hopkins University
12 Dweik, R.A. and McCarthy, K., "Pulmonary Function Testing", emedicine at: http://www.emedicine.com/med/topic2972.htm#target1
13 "Tracheomalacia": Medline Plus Health Information (Encyclopedia) http://www.nlm.nih.gov/medlineplus/ency/article/001084.htm
14 Polsdorfer, J. Ricker, Relapsing Polychondritis. Health Topics A-Z; secondary sourcesGilliland, Bruce C. "Relapsing Polychondritis and Other Arthritides." Harrison's Principles of Internal Medicine, edited by Antony S. Fauci, et al. New York: McGraw-Hill, 1998.: Schumacher, H. Ralph. "Relapsing Polychondritis." Cecil Textbook of Medicine, edited by J. Claude Bennett and Fred Plum. Philadelphia: W. B. Saunders, 1996.
15Tso, A.S, Chung, HS, Wu, C-Y, Li, J-Y, Hong, C-L, Yang, M-W, Lui, P-W., Anesthetic Management of a Patient with Relapsing Polychondritis-A Case Report. Acta Anaesthesiol Sin 39: 189-194, 2001.
16Waheed, N K, Relapsing Polychondritis
17Wyser, C, Stulz, P, Soler, M: Prospective evaluation of an algorithm for the functional assessment of lung resection candidates. Am J. Respir Crit Care Med 1999; 159-5 Pt 1_: 1450-6, (second sourced from reference 12 above)
18Warnock M.L. and McCowin, M.J. "Emphysema" (Overview of Pulmonary Pathology) in Practical Pathology of Chest Disease--Case Studies
19Introduction to Clinical Radiology, Radiology Pathology - BMS 5191, University of Florida College of Medicine
20Michael P. D'Alessandro, M.P and Yasayuki Kurihara,Y., Emphysema (Classic) on CXR, University of Iowa
21Hypertexts for Biomedical Sciences, Colorado State University, Pathophysiology
22Gal, T. J. "Respiratory Gas Exchange" in Respiratory Physiology in Anesthetic Practice, p. 133, William and Wilkins, Baltimore, 1991.
23Bouchey, H.A. "Drugs Used in Asthma" in Basic and Clinical Pharmacolgy (B.G. Katzung, ed.), pp. 338-339, 8th Edition, Lange Medical Group / McGraw Hill, New York, 2001.
24Jaksha, M and Wallis, J. (case authors) "Functional Right-to-Left Shunting" Division of Nuclear Medicine, Mallinkrodt Institute of Radiology, Washington University Medical Center http://gamma.wustl.edu/vq030te167.html
25Tisi, GN: Pulmonary physiology in clinical medicine, Baltimore, 1980, Williams and Wilkins (second sourced from ref. 1)